Business Model
Rapid development of new drugs by efficient management model
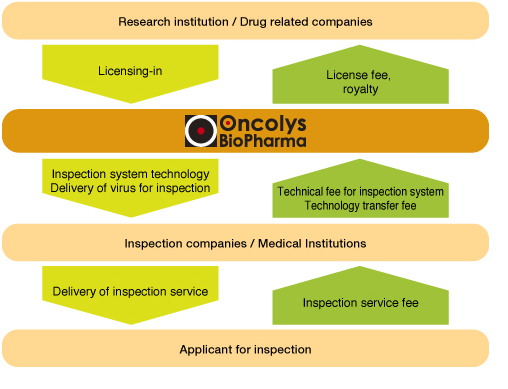
We are conducting the drug development by 'Planning & Operation,' that is so called fabless management, planning the new drug development, and outsourcing manufacture, pre-clinical and clinical study of such drug. This enables to optimize the costs and expenses of the development and shorten the period of time of the research and development.
Stable revenue base consisting of pharmaceutical and diagnostic businesses
In pharmaceutical business, we obtain a license of drug candidates targeting intractable diseases such as cancer and serious infectious diseases from research institute of academia and companies, and we are outsourcing the clinical development at early stage. Our earnings model is as follows. After obtaining Proof of Concept (POC) that is initial evaluation of product value of such drug candidate, we grant a license of the drug candidate to major pharmaceutical or biotech companies, and receive upfront payment, milestone payment in accordance with the progress of the development, and running royalties after the launch of the product. In diagnostic business, we provide clinical testing companies and medical institutions with inspection system as inspection unit using our virus reagents, and receive payments such as technology license fee or technology transfer fee. We aim to establish a continuous, stable and profitable business structure.
Business model in pharmaceutical business
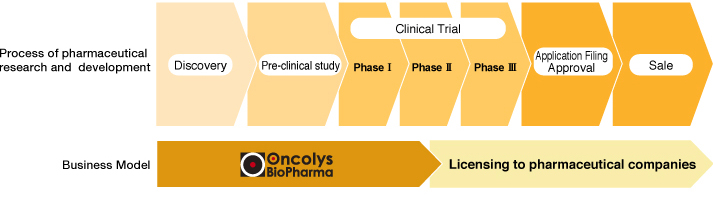
Business model in diagnostic business